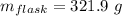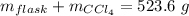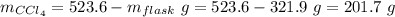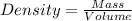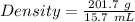Chemistry, 27.11.2019 02:31, Jakyramason
Aflask with a mass of 321.9 g is filled with 15.7 ml of carbon tetrachloride. the mass of the flask and carbon tetrachloride is found to be 523.6 g. from this information, calculate the density of carbon tetrachloride. according to this problem, the density of ccl4 is answer g/ml.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, lindseyklewis1p56uvi
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1



Chemistry, 23.06.2019 10:30, oglejack6138
How is it possible for someone to put an ear to a wall and hear someone in the next room? a. sound waves can travel though solids. b. the waves travel from room to room via air. c. there must be some air in the wall so the sound can travel through it. d. sound waves change to electromagnetic waves and then back again.
Answers: 1
Do you know the correct answer?
Aflask with a mass of 321.9 g is filled with 15.7 ml of carbon tetrachloride. the mass of the flask...
Questions in other subjects:

Mathematics, 16.04.2020 16:24

Mathematics, 16.04.2020 16:24


Mathematics, 16.04.2020 16:24

Health, 16.04.2020 16:24

Mathematics, 16.04.2020 16:24

Mathematics, 16.04.2020 16:24


Mathematics, 16.04.2020 16:24