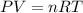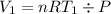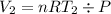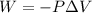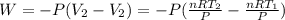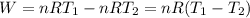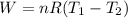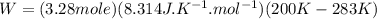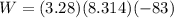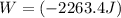Chemistry, 26.11.2019 23:31, parkerwallace04
Acylinder with a moveable piston contains 92g of nitrogen. the external pressure is constant at 1.00 atm. the initial temperature is 200k. when the temperature is increased by 83 k, by taking it out of the freezer, the volume will increase, according to the ideal gas law. calculate the work for this process. express your answer in j.

Answers: 2
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 07:30, tntaylor862
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1
Do you know the correct answer?
Acylinder with a moveable piston contains 92g of nitrogen. the external pressure is constant at 1.00...
Questions in other subjects:

History, 12.12.2019 20:31