
Chemistry, 26.11.2019 21:31, AshlynPlayz45
Consider the following reaction at constant pressure. use the information provided below to determine the value of δs at 473 k. predict whether or not this reaction will be spontaneous at this temperature.
4nh3 (g) + 3o2 (g) → 2n2 (g) + 6h2o (g) δh = –1267 kj

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:50, nnaomii
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% pb and 95.5 wt% sn? the atomic weights for pb and sn are 207.19 g/mol and 118.71 g/mol, respectively.(a) 2.6 at% pb and 97.4 at% sn(b) 7.6 at% pb and 92.4 at% sn(c)97.4 at% pb and 2.6 at% sn(d) 92.4 at% pb and 7.6 at% sn
Answers: 2


Chemistry, 22.06.2019 18:30, bibiansolis
The table lists the lattice energies of some compounds. compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf. the lattice energy increases as the cations get larger, as shown by lif and licl. the lattice energy decreases as cations get smaller, as shown by nacl and naf. the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 21:00, thebasedgodchri
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1
Do you know the correct answer?
Consider the following reaction at constant pressure. use the information provided below to determin...
Questions in other subjects:

Mathematics, 22.06.2019 15:40

History, 22.06.2019 15:40



Biology, 22.06.2019 15:40




English, 22.06.2019 15:40

Mathematics, 22.06.2019 15:40


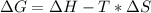
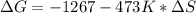 :
: ![\Delta S0[/tex}Back to this expression: [tex]\Delta G= -1267 - 473 K* \Delta S](/tpl/images/0392/0613/808f0.png)




