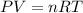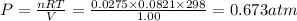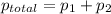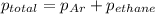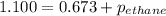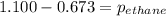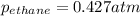Chemistry, 26.11.2019 06:31, Aviannakelly
A1.00 l flask is filled with 1.10 g of argon at 25 ∘c. a sample of ethane vapor is added to the same flask until the total pressure is 1.100 atm .1. part awhat is the partial pressure of argon, par, in the flask? express your answer to three significant figures and include the appropriate units.2. part bwhat is the partial pressure of ethane, pethane, in the flask? express your answer to three significant figures and include the appropriate units.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, Gghbhgy4809
What pressure will be exerted by 0.675 moles of a gas at 25*c if it is in a 0.750-l container?
Answers: 1



Chemistry, 22.06.2019 21:50, BookandScienceNerd
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
Do you know the correct answer?
A1.00 l flask is filled with 1.10 g of argon at 25 ∘c. a sample of ethane vapor is added to the same...
Questions in other subjects:


Mathematics, 28.02.2020 03:44

Mathematics, 28.02.2020 03:44

Mathematics, 28.02.2020 03:44


Biology, 28.02.2020 03:44


Chemistry, 28.02.2020 03:44

Mathematics, 28.02.2020 03:44