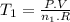Chemistry, 26.11.2019 06:31, kinziemadison12
Achemist prepares a sample of helium gas at a certain pressure, temperature and volume and then removes all but a fourth of the gas molecules (only a fourth remain). how must the temperature be changed (as a multiple of t1) to keep the pressure and the volume the same? a. t2=1/16t1b. t2=2t1c. t2=16t1d. t2= 1/2t1e. t2=4t1f. none of theseg. t2=1/4t1

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, JJlover1892
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 16:00, corcoranrobert1959
How do dying stars contribute to the formation of planets
Answers: 1

Chemistry, 22.06.2019 19:20, evansh78
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 23.06.2019 04:00, zakarycrane8101
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 3
Do you know the correct answer?
Achemist prepares a sample of helium gas at a certain pressure, temperature and volume and then remo...
Questions in other subjects:

Mathematics, 19.05.2021 20:50

Mathematics, 19.05.2021 20:50


Health, 19.05.2021 20:50