Chemistry, 25.11.2019 19:31, klocke2001
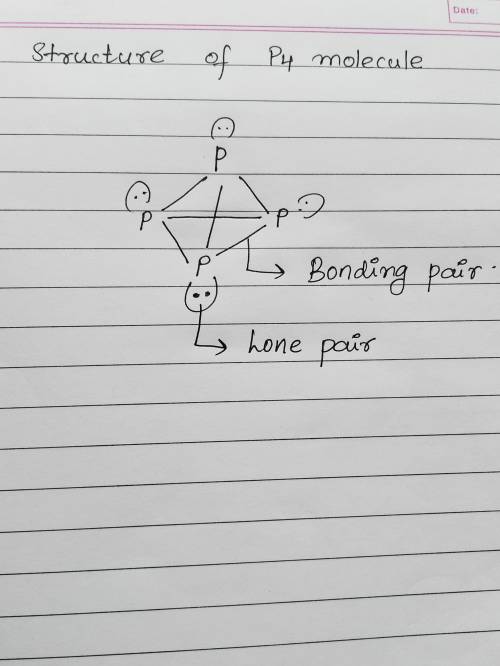
White phosphorus is composed of tetrahedral molecules of p4 in which every p atom is connected to three other p atoms. in the lewis structure of p4, there are 1. 6 bonding pairs and 4 lone pairs of electrons. 2. 6 bonding pairs and no lone pairs of electrons. 3. 5 bonding pairs and 4 lone pairs of electrons. 4. 3 bonding pairs and 4 lone pairs of electrons. 5. 6 bonding pairs and 2 lone pairs of electrons.

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 10:30, kluckey3426
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1
Do you know the correct answer?
White phosphorus is composed of tetrahedral molecules of p4 in which every p atom is connected to th...
Questions in other subjects:

History, 28.08.2019 07:10

English, 28.08.2019 07:10

Health, 28.08.2019 07:10

Biology, 28.08.2019 07:10

English, 28.08.2019 07:10

Mathematics, 28.08.2019 07:10

Computers and Technology, 28.08.2019 07:10


Mathematics, 28.08.2019 07:10