Someone me with 84 and explain got to do it : ((
...

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, penny3109
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1


Chemistry, 22.06.2019 20:30, kittybatch345
Is a chemical message sent by another individual.
Answers: 1

Chemistry, 22.06.2019 23:30, shukriabdisabrie
Match each statement with the state of matter it describes
Answers: 3
Do you know the correct answer?
Questions in other subjects:

Mathematics, 21.05.2020 02:02







Mathematics, 21.05.2020 02:02


Biology, 21.05.2020 02:02



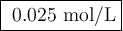
![K_{sp} =\text{[Ca$^{2+}$][OH$^{-}$]}^{2} = x\times (2x)^{2} = 4x^{3} = 6.5 \times 10^{-6}\\x^{3} = \dfrac{6.5\times 10^{-6}} {4} =1.62 \times 10^{-6}\\\\x = \sqrt[3]{1.62 \times 10^{-6}} = \textbf{0.025 mol/L}\\\text{The concentration of Ca$^{2+}$ ions is $\large \boxed{\textbf{ 0.025 mol/L}}$}](/tpl/images/0388/6219/c09ce.png)




