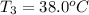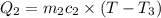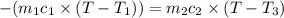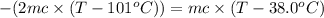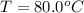Chemistry, 23.11.2019 00:31, sidallen05
One piece of copper jewelry at 101°c has twice the mass of another piece, which is at 38.0°c. both pieces are placed inside a calorimeter of negligible heat capacity. what is the final temperature inside the calorimeter (c of copper=0.387 j/g. k)?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:50, ayoismeisjjjjuan
Suppose you got a low yield of benzoin from your benzoin condensation reaction and thus only have 0.300 g of benzoin to use as the starting material for this reaction. how much concentrated nitric acid should you add? (concentrated nitric acid is 15.8 m). write your answer in the form x. xx ml
Answers: 1

Chemistry, 22.06.2019 15:20, mydoggy152
Fossil fuels are organic compounds that are made from
Answers: 1


Chemistry, 23.06.2019 10:30, fatheadd2007
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1
Do you know the correct answer?
One piece of copper jewelry at 101°c has twice the mass of another piece, which is at 38.0°c. both p...
Questions in other subjects:










Arts, 08.11.2019 04:31


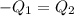
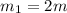
 = 0.387 J/g.K
= 0.387 J/g.K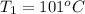
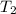 =T
=T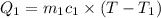
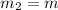
 = 0.387 J/g.K
= 0.387 J/g.K