
Chemistry, 21.11.2019 20:31, ilovewaffles70
Which statement best explains why the polarity of a h2o molecule differs from the polarity of a co2 molecule? the bond strength is greater in co2. oxygen is more electronegative than carbon. the central atom in h2o has lone pair electrons and the central atom in co2 does not.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, queenkimm26
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 23.06.2019 06:10, tammydbrooks43
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1

Chemistry, 23.06.2019 07:50, dootdootkazoot
What is the significance sodium hydroxide and hydrochloric acid
Answers: 1
Do you know the correct answer?
Which statement best explains why the polarity of a h2o molecule differs from the polarity of a co2...
Questions in other subjects:

Social Studies, 30.06.2019 17:00

Mathematics, 30.06.2019 17:00

Mathematics, 30.06.2019 17:00


Mathematics, 30.06.2019 17:00


Mathematics, 30.06.2019 17:00


Mathematics, 30.06.2019 17:00


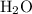 molecule differs from the polarity of
molecule differs from the polarity of 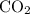 molecule, because Oxygen is more electronegative than carbon.
molecule, because Oxygen is more electronegative than carbon.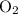 is 3.44.
is 3.44.



