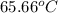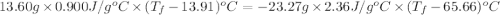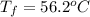Chemistry, 21.11.2019 05:31, tabocampos1414
A13.60-g block of solid aluminum at 13.91 °c is immersed in a 23.27-g pool of liquid ethylene glycol with a temperature of 65.66 °c. when thermal equilibrium is reached, what is the temperature of the aluminum and ethylene glycol? specific heat capacities: lead = 0.159 j/g °c; ethylene glycol = 2.36 j/g ° °c

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 22.06.2019 19:40, trodgers0202
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests. which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3

Chemistry, 23.06.2019 13:00, journeyburks07
The gram molecular mass or co2 is the same as the gram molecular mass of
Answers: 2
Do you know the correct answer?
A13.60-g block of solid aluminum at 13.91 °c is immersed in a 23.27-g pool of liquid ethylene glycol...
Questions in other subjects:


Mathematics, 11.05.2021 20:40







English, 11.05.2021 20:40

English, 11.05.2021 20:40


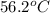
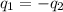
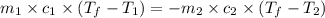
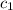 = specific heat of aluminum =
= specific heat of aluminum = 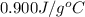
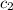 = specific heat of ethylene glycol =
= specific heat of ethylene glycol = 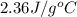
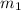 = mass of aluminum = 13.60 g
= mass of aluminum = 13.60 g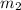 = mass of ethylene glycol = 23.27 g
= mass of ethylene glycol = 23.27 g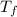 = final temperature of aluminum and ethylene glycol = ?
= final temperature of aluminum and ethylene glycol = ?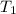 = initial temperature of aluminium =
= initial temperature of aluminium = 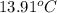
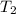 = initial temperature of ethylene glycol =
= initial temperature of ethylene glycol =