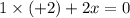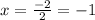Chemistry, 21.11.2019 04:31, rayniqueamee2002
Consider the lattice energies of the following group 2a compounds: beh2, 3205 kj/mol; mgh2, 2791 kj/mol; cah2, 2410 kj/mol; srh2, 2250 kj/mol; bah2, 2121 kj/mol. part a what is the oxidation number of h in these compounds?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, HHHHHHHHHMMMMMMMMM
About 70 percent of the earth's surface is water-covered, and about 96.5 percent of all earth's water is salt water. identify the watery feature on earth that is made of freshwater rather than salt water. a) bay b) glacier c) ocean d) sea it is not incomplete this is the true question
Answers: 1

Chemistry, 22.06.2019 20:00, 20calzoy
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 22.06.2019 21:30, leenzazou587
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
Do you know the correct answer?
Consider the lattice energies of the following group 2a compounds: beh2, 3205 kj/mol; mgh2, 2791 k...
Questions in other subjects:

History, 19.03.2020 23:57

Chemistry, 19.03.2020 23:57


Biology, 19.03.2020 23:57

Social Studies, 19.03.2020 23:57



Mathematics, 19.03.2020 23:57


Mathematics, 19.03.2020 23:57


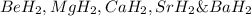 :
: