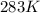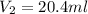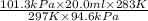Chemistry, 21.11.2019 03:31, Kalistaaaaaaa
Asample of oxygen gas in one container has a volume of 20.0mililiter at 297 k and 101.3 kpa. the entire sample is transferred to another container where the temperature is 283 k and the pressure is 94.6 kpa. which is correct numerical setup for calculating the new volume of this sample of oxygen gas
show work

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, malenacastillo4887
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 18:00, jessicannoh5965
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1

Chemistry, 22.06.2019 21:00, ciel8809
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1
Do you know the correct answer?
Asample of oxygen gas in one container has a volume of 20.0mililiter at 297 k and 101.3 kpa. the ent...
Questions in other subjects:

Mathematics, 18.07.2019 03:30


English, 18.07.2019 03:30

Mathematics, 18.07.2019 03:30

Mathematics, 18.07.2019 03:30

Chemistry, 18.07.2019 03:30


English, 18.07.2019 03:30



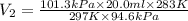
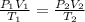
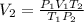
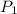 = initial pressure of gas = 101.3 kPa
= initial pressure of gas = 101.3 kPa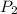 = final pressure of gas = 94.6 kPa
= final pressure of gas = 94.6 kPa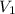 = initial volume of gas = 20.0 ml
= initial volume of gas = 20.0 ml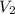 = final volume of gas = ?
= final volume of gas = ?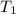 = initial temperature of gas =
= initial temperature of gas = 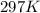
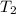 = final temperature of gas =
= final temperature of gas =