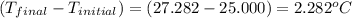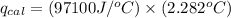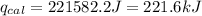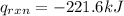Chemistry, 21.11.2019 01:31, Deadpool9609
4.41 g of propane gas (c3h8) is injected into a bomb calorimeter and ignited with excess oxygen, according to the reaction below. the calorimeter (including the water) has a heat capacity of 97.1 kj/°c. c3h8(g) + 5 o2(g) 3 co2(g) + 4 h2o() (a) if the temperature rose from 25.000°c to 27.282°c, what is the heat of the reaction, qrxn?

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 11:30, elijah1090
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Do you know the correct answer?
4.41 g of propane gas (c3h8) is injected into a bomb calorimeter and ignited with excess oxygen, acc...
Questions in other subjects:

English, 05.10.2020 15:01


History, 05.10.2020 15:01

Mathematics, 05.10.2020 15:01

History, 05.10.2020 15:01

Spanish, 05.10.2020 15:01


Arts, 05.10.2020 15:01



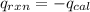
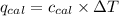
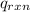 = heat released by the reaction = ?
= heat released by the reaction = ?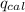 = heat absorbed by the calorimeter
= heat absorbed by the calorimeter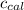 = specific heat of calorimeter =
= specific heat of calorimeter = 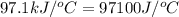
 = change in temperature =
= change in temperature =