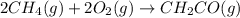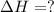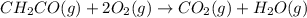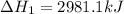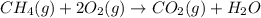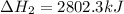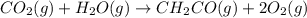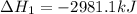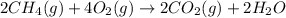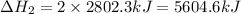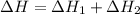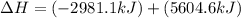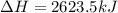Chemistry, 20.11.2019 06:31, treyhunt5362
The mea sured enthalpy change for burning ketene (ch2co) ch2co(g) 1 2 o2(g)88n 2 co2(g) 1 h2o(g) is dh1 5 2981.1 kj at 25°c. the enthalpy change for burning methane ch4(g) 1 2 o2(g)88n co2(g) 1 2 h2o(g) is dh2 5 2802.3 kj at 25°c. calculate the enthalpy change at 25°c for the reaction 2 ch4(g) 1 2 o2(g)88n ch2co(g) 1 3 h2o(g)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2


Chemistry, 22.06.2019 20:10, jakhunter354
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3
Do you know the correct answer?
The mea sured enthalpy change for burning ketene (ch2co) ch2co(g) 1 2 o2(g)88n 2 co2(g) 1 h2o(g) is...
Questions in other subjects:



Mathematics, 29.09.2019 19:20

Mathematics, 29.09.2019 19:20


English, 29.09.2019 19:20

French, 29.09.2019 19:20


Mathematics, 29.09.2019 19:20