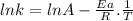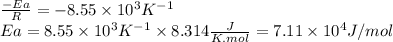The rate constant of a reaction is measured at different temperatures. a plot of the natural log of the rate constant as a function of the inverse of the temperature (in kelvins) yields a straight line with a slope of −8.55×103 k−1. what is the activation energy (ea) for the reaction?

Answers: 3
Other questions on the subject: Chemistry



Do you know the correct answer?
The rate constant of a reaction is measured at different temperatures. a plot of the natural log of...
Questions in other subjects:



Mathematics, 04.12.2021 21:40


Social Studies, 04.12.2021 21:40