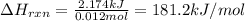Chemistry, 19.11.2019 00:31, keyonaemanieevans
Manganese reacts with hydrochloric acid to produce manganese(ii) chloride and hydrogen gas. mn(s)+2hcl(aq) ? mncl2(aq)+h2(g)
part a when 0.650g mn is combined with enough hydrochloric acid to make 100.0 ml of solution in a coffee-cup calorimeter, all of the mn reacts, raising the temperature of the solution from 23.0? c to 28.2? c. find ? hrxn for the reaction as written. (assume that the specific heat capacity of the solution is 4.18 j/g? c and the density is 1.00 g/ml.) express your answer using three significant figures. ? hrxn = kj

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, nothingworksoutforme
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 06:30, noathequeen
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 21:20, jordan2875
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1
Do you know the correct answer?
Manganese reacts with hydrochloric acid to produce manganese(ii) chloride and hydrogen gas. mn(s)+2h...
Questions in other subjects:





English, 06.05.2020 03:27


Mathematics, 06.05.2020 03:27


Biology, 06.05.2020 03:27


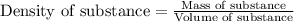
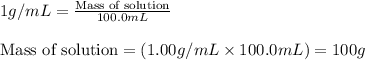
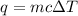
 = change in temperature =
= change in temperature = 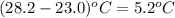
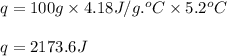
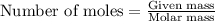
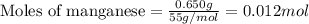
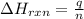
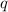 = amount of heat absorbed = 2173.6 J = 2.174 kJ (Conversion used: 1 kJ = 1000 J)
= amount of heat absorbed = 2173.6 J = 2.174 kJ (Conversion used: 1 kJ = 1000 J)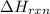 = enthalpy change of the reaction
= enthalpy change of the reaction