
Nh4+ (aq) + no2- (aq) → n2 (g) + h2o (l) experiment [nh4+]i [no2-]i initial rate (m/s) 1 0.24 0.10 7.2 x 10-4 2 0.12 0.10 3.6 x 10-4 3 0.12 0.15 5.4 x 10-4 4 0.12 0.12 4.3 x 10-4 first determine the rate law and rate constant. under the same initial conditions as in experiment 4, calculate [nh4+] at 274 seconds after the start of the reaction. in this experiment, both reactants are present at the same initial concentration. the units should be m, and should be calculated to three significant figures.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, mimireds5419
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3



Chemistry, 22.06.2019 19:40, jholland03
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1
Do you know the correct answer?
Nh4+ (aq) + no2- (aq) → n2 (g) + h2o (l) experiment [nh4+]i [no2-]i initial rate (m/s) 1 0.24 0.10 7...
Questions in other subjects:

Mathematics, 04.03.2021 18:10



Mathematics, 04.03.2021 18:10


English, 04.03.2021 18:10


Mathematics, 04.03.2021 18:10


Mathematics, 04.03.2021 18:10


![\text{Rate}=k[NH_4^+][NO_2^-]](/tpl/images/0380/3184/ed258.png)
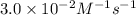
![[NH_4^+]](/tpl/images/0380/3184/5c46c.png) at 274 seconds after the start of the reaction is 0.0604 M
at 274 seconds after the start of the reaction is 0.0604 M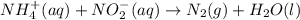
![\text{Rate}=k[NH_4^+]^a[NO_2^-]^b](/tpl/images/0380/3184/00132.png)
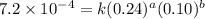 ....(1)
....(1)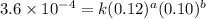 ....(2)
....(2)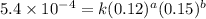 ....(3)
....(3)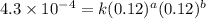 ....(4)
....(4)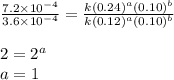
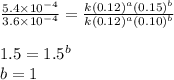
![\text{Rate}=k[NH_4^+]^1[NO_2^-]^1](/tpl/images/0380/3184/43151.png)
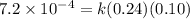
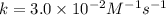
![[NO_2^-]](/tpl/images/0380/3184/10a69.png) are same and the reaction is 1st order for both.
are same and the reaction is 1st order for both.![kt=\frac{1}{[A_t]}-\frac{1}{[A_o]}](/tpl/images/0380/3184/ccade.png)
![[A_t]](/tpl/images/0380/3184/5262c.png) = final concentration = ?
= final concentration = ?![[A_o]](/tpl/images/0380/3184/dc622.png) = initial concentration = 0.12 M
= initial concentration = 0.12 M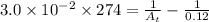
![[A_t]=0.0604M](/tpl/images/0380/3184/69b49.png)




