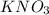Which of the following gives the balanced equation for this reaction?
k3po4 + ca(no3)2 → ca3...

Which of the following gives the balanced equation for this reaction?
k3po4 + ca(no3)2 → ca3(po4)2 + kno3
a) kpo4 + cano3 + kno3
b) k3po4 + ca(no3)2 → ca3(po4)2 + 3kno3
c) 2k3po4 + ca(no3)2 → ca3(po4)2 + 6kno3
d) 2k3po4 + 3ca(no3)2 → ca3(po4)2 + 6kno3
e) k3po4 + ca(no3)2 → ca3(po4)2 + kno3

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, hala201490
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 13:00, torigirl4126
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3

Chemistry, 22.06.2019 21:50, isabel81ie
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l. s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2
Do you know the correct answer?
Questions in other subjects:







Mathematics, 01.08.2019 15:30

English, 01.08.2019 15:30


Chemistry, 01.08.2019 15:30


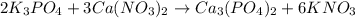
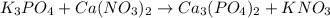
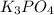 , coefficient 3 is put before the
, coefficient 3 is put before the 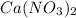 and 6 is put before the
and 6 is put before the