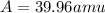Three isotopes of argon occur in nature – 36 18ar, 38 18ar, 40 18ar. calculate the average atomic mass of argon to two decimal places, given the following relative atomic masses and the abundances of each of the isotopes: argon36 (35.97 amu; 0.337%), argon-38 (37.96 amu; 0.063%), argon-40 (39.96 amu; 99.600%). 1. 119.89 amu 2. 39.95 amu 3. 39.96 amu 4. 35.97 amu 5. none of these 6. 37.96 amu 7. 37.95 amu 8. 35.96 amu

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, babygirl2984
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1


Chemistry, 22.06.2019 10:30, cheyennecarrillo14
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1
Do you know the correct answer?
Three isotopes of argon occur in nature – 36 18ar, 38 18ar, 40 18ar. calculate the average atomic ma...
Questions in other subjects:

Mathematics, 04.01.2021 05:50



English, 04.01.2021 05:50

Mathematics, 04.01.2021 05:50




Mathematics, 04.01.2021 06:00

Chemistry, 04.01.2021 06:00






![A=\sum[(35.97\times 3.37\times 10^{-3})+(37.96\times 6.3\times 10^{-4})+(39.96\times 0.996)]](/tpl/images/0377/0178/15521.png)