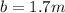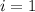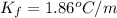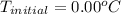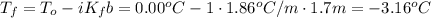Chemistry, 15.11.2019 20:31, elizabethhubbe
What would be the freezing point of a 1.7 mole aqueous ethylene glycol solution? the freezing point depression constant for water = 1.86 degrees celsius per mole.
a) 3.2°c
b) – 1.1°c
c) 0.0°c
d) – 3.2°c

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 11:40, Wemaybewrong
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 12:30, fvmousdiana
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3
Do you know the correct answer?
What would be the freezing point of a 1.7 mole aqueous ethylene glycol solution? the freezing point...
Questions in other subjects:






Business, 16.01.2020 17:31





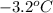
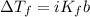
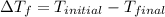 is the change in the freezing point of the solvent given its initial and final freezing point temperature values;
is the change in the freezing point of the solvent given its initial and final freezing point temperature values; is the van 't Hoff factor (i = 1 for non-electrolyte solutes and i depends on the number of moles of ions released per mole of ionic salt);
is the van 't Hoff factor (i = 1 for non-electrolyte solutes and i depends on the number of moles of ions released per mole of ionic salt); is the freezing point depression constant for the solvent;
is the freezing point depression constant for the solvent;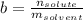 is molality of the solute, defined as a ratio between the moles of solute and the mass of solvent (in kilograms).
is molality of the solute, defined as a ratio between the moles of solute and the mass of solvent (in kilograms).