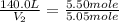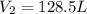Chemistry, 14.11.2019 04:31, jahnoibenjamin
A140.0 l weather balloon filled with 5.50 moles of helium has a small leak. if the helium leaks at a rate of 10.0 mmol/hr, what is the volume of the balloon after 45.0 hours?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kiki197701
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3

Chemistry, 22.06.2019 08:30, Apple557
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 17:10, sophiaa23
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1

Chemistry, 23.06.2019 00:50, kaseywright3418
Which statement would indicate the presence of an acid
Answers: 3
Do you know the correct answer?
A140.0 l weather balloon filled with 5.50 moles of helium has a small leak. if the helium leaks at a...
Questions in other subjects:

Physics, 22.10.2019 15:00

History, 22.10.2019 15:00



History, 22.10.2019 15:00



Chemistry, 22.10.2019 15:00


Mathematics, 22.10.2019 15:00


![\frac{45.0hr]{1hr}\times 10.0=450mmol=0.450mole](/tpl/images/0373/5494/4216a.png)
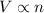
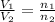
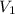 = initial volume of gas = 140.0 L
= initial volume of gas = 140.0 L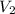 = final volume of gas = ?
= final volume of gas = ?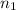 = initial moles of gas = 5.50 mole
= initial moles of gas = 5.50 mole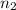 = final moles of gas = 5.05 mole
= final moles of gas = 5.05 mole