p4(s)+10hclo(aq)+6h2o(l)? 4h3po4(aq)+10hcl(aq)

Chemistry, 13.11.2019 00:31, jojo10092003
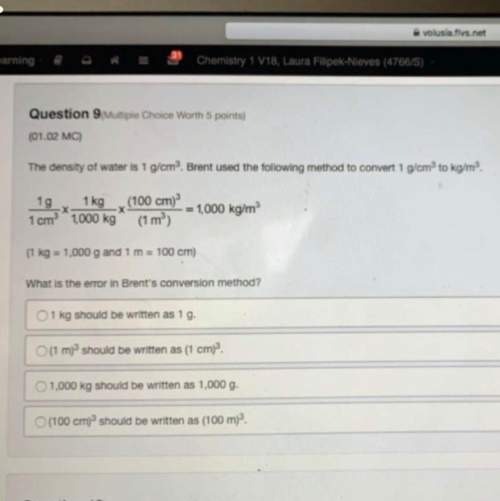
Which of the following are redox reactions?
p4(s)+10hclo(aq)+6h2o(l)? 4h3po4(aq)+10hcl(aq)
br2(l)+2k(s)? 2kbr(s)
ch3ch2oh(l)+3o2(g)? 3h2o(l)+2co2(g)
zncl2(aq)+2naoh(aq)? zn(oh)2(s)+2nacl(aq)
check all that apply.
p4(s) + 10hclo(aq) + 6h2o(l) ? 4h3po4(aq) + 10hcl(aq)
br2(l) + 2k(s) ? 2kbr(s)
ch3ch2oh(l) + 3o2(g) ? 3h2o(l) + 2co2(g)
zncl2(aq) + 2naoh(aq) ? zn(oh)2(s) + 2nacl(aq)
part b
for those reactions that are redox, indicate which elements are oxidized.
express your answers as chemical symbols separated by a commas.
part c
for those reactions that are redox, indicate which elements are reduced.
express your answers as chemical symbols separated by a commas.
part d
for those reactions that are not redox, indicate whether they are precipitation or neutralization reactions.
~
when solutions of silver nitrate and calcium chloride are mixed, silver chloride precipitates out of solution according to the equation
2agno3(aq)+cacl2(aq)? 2agcl(s)+ca(no3)2(aq)
part a
what mass of silver chloride can be produced from 1.51l of a 0.201m solution of silver nitrate?
express your answer with the appropriate units.
the reaction described in part a required 3.51l of calcium chloride. what is the concentration of this calcium chloride solution?
express your answer with the appropriate units.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 18:50, christhegreat1
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 22.06.2019 19:30, Adrian12313
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1


Chemistry, 22.06.2019 22:30, darkshaders11
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1
Do you know the correct answer?
Which of the following are redox reactions?
p4(s)+10hclo(aq)+6h2o(l)? 4h3po4(aq)+10hcl(aq)
p4(s)+10hclo(aq)+6h2o(l)? 4h3po4(aq)+10hcl(aq)
Questions in other subjects:






Biology, 08.10.2021 06:50

Mathematics, 08.10.2021 06:50

Chemistry, 08.10.2021 06:50

Mathematics, 08.10.2021 06:50

Mathematics, 08.10.2021 06:50