
Chemistry, 13.11.2019 00:31, staz13wiggins
The following sequence of reactions occurs in the commercial production of aqueous nitric acid. (the overall industrial process produces excess no.) 4 nh3(g) + 5 o2(g) → 4 no(g) + 6 h2o(l) δh = −1166.0 kj/mol (1) 2 no(g) + o2(g) → 2 no2(g) δh = −116.2 kj/mol (2) 3 no2(g) + h2o(l) → 2 hno3(aq) + no(g) δh = −137.3 kj/mol (3) determine the total energy change (in kj) for the production of one mole of aqueous nitric acid by this process.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, ian2006huang
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 06:10, andybiersack154
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1


Chemistry, 22.06.2019 20:00, ahnorthcutt4965
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1
Do you know the correct answer?
The following sequence of reactions occurs in the commercial production of aqueous nitric acid. (the...
Questions in other subjects:





Physics, 24.02.2021 16:30

Mathematics, 24.02.2021 16:30

English, 24.02.2021 16:30

History, 24.02.2021 16:30



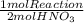 = 709,8 kJ
= 709,8 kJ



