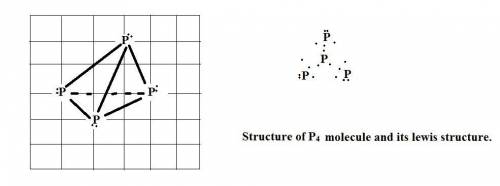
Chemistry, 12.11.2019 21:31, jeremyrs101
The standard state of phosphorus at 25∘c is p4. this molecule has four equivalent p atoms, no double or triple bonds, and no expanded octets. draw its lewis structure. draw the molecule by placing atoms on the grid and connecting them with bonds. include all non-bonding electrons.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, mgnbrnne
Two nitro no2 groups are chemically bonded to a patch of surface. they can't move to another location on the surface, but they can rotate (see sketch at right). it turns out that the amount of rotational kinetic energy each no2 group can have is required to be a multiple of ε, where =ε×1.010−24 j. in other words, each no2 group could have ε of rotational kinetic energy, or 2ε, or 3ε, and so forth — but it cannot have just any old amount of rotational kinetic energy. suppose the total rotational kinetic energy in this system is initially known to be 32ε. then, some heat is removed from the system, and the total rotational kinetic energy falls to 18ε. calculate the change in entropy. round your answer to 3 significant digits, and be sure it has the correct unit symbol.
Answers: 2

Chemistry, 22.06.2019 08:40, jaueuxsn
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

Chemistry, 22.06.2019 09:00, wkalpakchi
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1
Do you know the correct answer?
The standard state of phosphorus at 25∘c is p4. this molecule has four equivalent p atoms, no double...
Questions in other subjects:


History, 17.01.2020 07:31

Chemistry, 17.01.2020 07:31

Geography, 17.01.2020 07:31

Mathematics, 17.01.2020 07:31


Mathematics, 17.01.2020 07:31



History, 17.01.2020 07:31


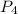 molecule.the
molecule.the 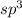 hybridized phosphorus atom lies in the corner of the regular tetrahedron. Each phosphorus atom is connected to three other phosphorus atoms.
hybridized phosphorus atom lies in the corner of the regular tetrahedron. Each phosphorus atom is connected to three other phosphorus atoms.