Chemistry, 12.11.2019 21:31, lizbeth232001
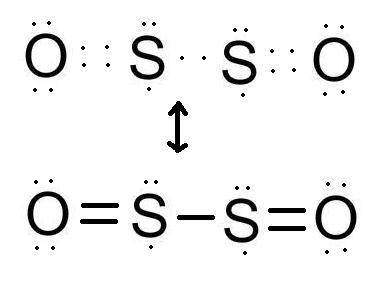
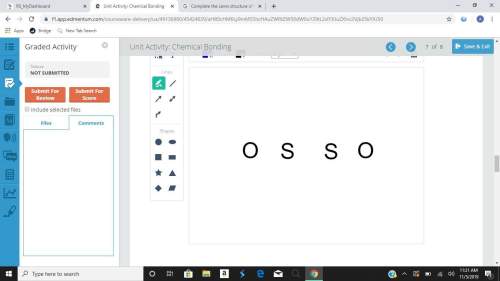
Complete the lewis structure of the molecule. draw the missing dots and dashes representing the valence electrons in the molecule.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, kathleensumter4913
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 22.06.2019 03:50, Pizzapegasus1
Express the following number in scientific notation. 0.026890 =
Answers: 1

Chemistry, 22.06.2019 04:50, aletadaboss
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1
Do you know the correct answer?
Complete the lewis structure of the molecule. draw the missing dots and dashes representing the vale...
Questions in other subjects:

History, 04.01.2020 00:31



History, 04.01.2020 00:31



Biology, 04.01.2020 00:31




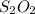 is shown below.
is shown below.