
250 milliliters of a concentrated iron (ii) ion solution are sitting on the lab bench in need of analysis to determine its exact concentration of fe2+ ions. as part of the analysis a 15 ml sample of this solution was diluted to a final volume of 100 ml. the iron (ii) ion concentration of the diluted solution was found to be 1.35 x 10-3 mg/ml fe2+. what was the total number of milligrams of iron (ii) that were contained in the original solution?
note that this question is not asking for how many mg/ml of iron (ii) where in the original solution, but the total mass of iron contained in the original solution. hint: you must first calculate the mg/ml of iron (ii) in the original solution.

Answers: 3
Similar questions

Mathematics, 07.07.2019 13:00, lovely222
Answers: 1

Mathematics, 02.08.2019 00:30, travyse
Answers: 1

Chemistry, 27.08.2019 18:30, ahmadfarriz4088
Answers: 1

Chemistry, 17.09.2019 22:30, alizeleach0123
Answers: 1
Do you know the correct answer?
250 milliliters of a concentrated iron (ii) ion solution are sitting on the lab bench in need of ana...
Questions in other subjects:


Physics, 17.12.2020 22:50


Mathematics, 17.12.2020 22:50

Mathematics, 17.12.2020 22:50






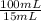 = 9,00x10⁻³ mg/mL
= 9,00x10⁻³ mg/mL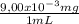 = 2,25 mg of Fe²⁺
= 2,25 mg of Fe²⁺




