
Chemistry, 12.11.2019 02:31, rubincain203
Write a balanced equation for the combustion of c7h16(l) (heptane) -- i. e. its reaction with o2(g) forming the products co2(g) and h2o(l). given the following standard heats of formation: δhf° of co2(g) is -393.5 kj/mol δhf° of h2o(l) is -286 kj/mol δhf° of c7h16(l) is -187.8 kj/mol what is the standard heat of reaction (δh°) for the combustion reaction of c7h16(l)? -4854.7 kj calculate the difference, δh-δe=δ(pv) for the combustion reaction of 1 mole of heptane. (assume standard state conditions and 298 k for all reactants and products.)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, aeverettpdzrvo
The most efficient way to establish the best possible economizer position is to measure
Answers: 1

Chemistry, 22.06.2019 06:00, kylieweeks052704
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 10:00, sdlesley66
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Do you know the correct answer?
Write a balanced equation for the combustion of c7h16(l) (heptane) -- i. e. its reaction with o2(g)...
Questions in other subjects:



Mathematics, 20.02.2020 17:42


Business, 20.02.2020 17:43






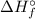 ) for: C₇H₁₆ (l) = -187.8 kJ/mol, O₂ (g) = 0 kJ/mol, CO₂ (g) = -393.5 kJ/mol, H₂O (l) = -286 kJ/mol
) for: C₇H₁₆ (l) = -187.8 kJ/mol, O₂ (g) = 0 kJ/mol, CO₂ (g) = -393.5 kJ/mol, H₂O (l) = -286 kJ/mol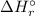 ) can be calculated by the Hess's law:
) can be calculated by the Hess's law:![\Delta H _{r}^{\circ } = \left [\sum \nu \cdot\Delta H _{f}^{\circ }(products) \right ] - \left [\sum \nu\cdot\Delta H _{f}^{\circ }(reactants) \right ]](/tpl/images/0369/9572/a156f.png)
 is the stoichiometric coefficient
is the stoichiometric coefficient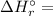
![\left [ 7\times \Delta H _{f}^{\circ }\left (CO_{2}\right )+ 8\times \Delta H _{f}^{\circ }\left (H_{2}O \right )\right ]](/tpl/images/0369/9572/3f95a.png)
![- \left [1\times \Delta H _{f}^{\circ }\left (C_{7}H_{16}\right ) +11\times \Delta H _{f}^{\circ }\left (O_{2} \right ) \right ]](/tpl/images/0369/9572/f3670.png)
![=\left [ 7\times \left (-393.5 kJ/mol \right )+ 8\times \left (-286 kJ/mol \right )\right ]](/tpl/images/0369/9572/7a112.png)
![-\left [1\times \left (-187.8 kJ/mol \right ) +11\times \left (0 kJ/mol \right ) \right ]](/tpl/images/0369/9572/be2b2.png)
![\Delta H _{r}^{\circ } = \left [ \left (-2754.5 \right )+ \left (-2288 \right )\right ]\left -[ \left (-187.8 \right ) +\left (0 \right )\right ]](/tpl/images/0369/9572/e3fd5.png)
![\Delta H _{r}^{\circ } = \left [ -5042.5 ]\left -[ -187.8] = \left ( -4854.7kJ \right )](/tpl/images/0369/9572/c52a2.png)




