
The overall kf for the complex ion ag(nh3)2+ is 1.7 x 107. the ksp for agi is 8.5 x 10-17. what is the molar solubility of agi in a solution that is 2.0 m in nh3? the overall kf for the complex ion ag(nh3)2+ is 1.7 x 107. the ksp for agi is 8.5 x 10-17. what is the molar solubility of agi in a solution that is 2.0 m in nh3? 1.3 x 10-3 8.4 x 10-5 5.8 x 10-12 1.5 x 10-9 7.6 x 10-5

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, melikefood01
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1

Chemistry, 22.06.2019 18:30, chinadoll24
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 22.06.2019 23:00, Mw3spartan17
What extra step distinguishes fermentation from glycolysis
Answers: 1
Do you know the correct answer?
The overall kf for the complex ion ag(nh3)2+ is 1.7 x 107. the ksp for agi is 8.5 x 10-17. what is t...
Questions in other subjects:



Mathematics, 02.09.2019 07:10

World Languages, 02.09.2019 07:10


Mathematics, 02.09.2019 07:10

Mathematics, 02.09.2019 07:10

Biology, 02.09.2019 07:10



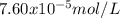
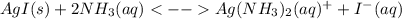
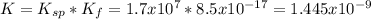
![K=\frac{[Ag(NH_3)_2^+][I^-]}{[NH_3]^2} \\K=\frac{x^2}{(2.0-2x)^2}\\\sqrt{K}= \sqrt{\frac{x^2}{(2.0-2x)^2}}\\2.0\sqrt{K}-2\sqrt{K}x-x=0\\7.60x10^{-5}-7.60x10^{-5}x-x=0\\x=\frac{7.60x10^{-5}}{1} =7.60x10^{-5}mol/L](/tpl/images/0367/6284/ce7cd.png)




