
Chemistry, 09.11.2019 05:31, elijahlylejamez45
Ammonia is produced commercially by the direct reaction of the elements. the formation of 1.00 moles of gaseous nh3 by this reaction releases 46.17 kj of heat. how much energy (in kj) is released, when 28.0 kg of hydrogen gas, h2, reacts in excess nitrogen gas, n2?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:40, gabrielolivas59
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3



Chemistry, 22.06.2019 21:50, SoccerAllStar2
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2
Do you know the correct answer?
Ammonia is produced commercially by the direct reaction of the elements. the formation of 1.00 moles...
Questions in other subjects:






Social Studies, 02.08.2019 13:30

Mathematics, 02.08.2019 13:30

Chemistry, 02.08.2019 13:30


History, 02.08.2019 13:30


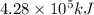 of heat energy will be released
of heat energy will be released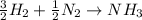
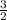 moles of
moles of 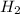 produces 1 mol of
produces 1 mol of 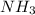 to release 46.17 kJ of heat.
to release 46.17 kJ of heat.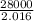 moles of
moles of 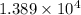 moles of
moles of 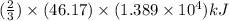 heat or
heat or 



