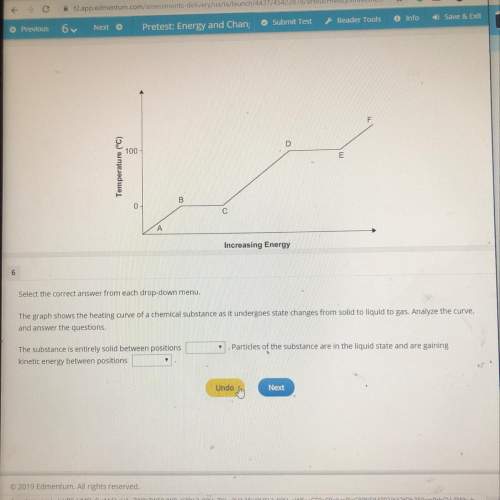
The graph shows the heating curve of a chemical substance as it undergoes state changes from solid to liquid to gas. analyze the curve,
and answer the questions.
the substance is entirely solid between positions
kinetic energy between positions
particles of the substance are in the liquid state and are gaining

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, poopybutt541
Avariable that is not being directly tested during an experiment should be
Answers: 1


Chemistry, 22.06.2019 14:30, KennyOaks6230
Which of the following units is not an official si unit
Answers: 1

Chemistry, 22.06.2019 20:10, jakhunter354
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3
Do you know the correct answer?
The graph shows the heating curve of a chemical substance as it undergoes state changes from solid t...
Questions in other subjects:

Mathematics, 26.06.2020 17:01




Mathematics, 26.06.2020 17:01

Mathematics, 26.06.2020 17:01

Mathematics, 26.06.2020 17:01

Social Studies, 26.06.2020 17:01


Mathematics, 26.06.2020 17:01