
Chemistry, 07.11.2019 21:31, BigGirlsTheBest
At a certain temperature, the equilibrium constant, kc, kc, for this reaction is 53.3. h2(g)+i2(g)↽−−⇀2hi(g)kc=53.3 h2(g)+i2(g)↽−−⇀2hi(g)kc=53.3 at this temperature, 0.400 mol h20.400 mol h2 and 0.400 mol i20.400 mol i2 were placed in a 1.00 l container to react. what concentration of hihi is present at equilibrium?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:40, petriajack8375
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3

Chemistry, 22.06.2019 04:00, fantasticratz2
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 08:30, lpssprinklezlps
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3

Chemistry, 22.06.2019 11:20, Jessicadiaz8602
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1
Do you know the correct answer?
At a certain temperature, the equilibrium constant, kc, kc, for this reaction is 53.3. h2(g)+i2(g)↽−...
Questions in other subjects:



Biology, 16.10.2019 23:00

Mathematics, 16.10.2019 23:00


English, 16.10.2019 23:00

Health, 16.10.2019 23:00


Mathematics, 16.10.2019 23:00


![K_{c} = \frac{[HI]^{2}}{[H_{2}][I_{2}]} = 53.3](/tpl/images/0364/2800/9c8ef.png) (2)
(2)![[H_{2}] = [I_{2}] = \frac{0.400 mol}{1 L} = 0.400 mol/L](/tpl/images/0364/2800/3a013.png)
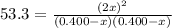



![[HI] = 2x = 2*0.31 mol/L = 0.62 mol/L](/tpl/images/0364/2800/ce8eb.png)
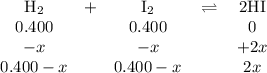
![K_{\text{c}} = \dfrac{\text{[HI]$^{2}$}}{\text{[H$_{2}$][I$_2$]}} = \dfrac{(2x)^{2}}{(0.400 - x)^{2}} = 53.3\\\\\begin{array}{rcl}\dfrac{(2x)^{2}}{(0.400 - x)^{2}} &=& 53.3\\ \dfrac{2x }{0.400 - x} & = & 7.301\\\\2x & = & 7.301(0.400 - x)\\2x & = & 2.920 - 7.301x\\9.301x & = & 2.920\\x & = & \dfrac{2.920}{9.301}\\\\x & = & \mathbf{0.3140}\\\end{array}](/tpl/images/0364/2800/cfef9.png)





