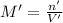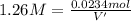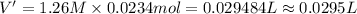Let us assume that cr(oh)3(s) is completely insoluble, which signifies that the precipitation reaction with naoh(aq) (presented in the transition) would go to completion.
cr3+(aq)+3naoh(aq) → cr(oh)3(s)+3na+(aq)
if you had a 0.600 l solution containing 0.0130 m of cr3+(aq), and you wished to add enough 1.26 m naoh(aq) toprecipitate all of the metal, what is the minimum amount of the naoh(aq) solution you would need to add? assume that the naoh(aq) solution is the only source of oh−(aq) for the precipitation.
express the volume to three significant figures and include the appropriate units.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 22:30, lanashanabJHsbd1099
Who discovered a pattern to the elements in 1869?
Answers: 1
Do you know the correct answer?
Let us assume that cr(oh)3(s) is completely insoluble, which signifies that the precipitation reacti...
Questions in other subjects:

History, 27.09.2019 07:10


English, 27.09.2019 07:10


Mathematics, 27.09.2019 07:10

Mathematics, 27.09.2019 07:10

English, 27.09.2019 07:10



English, 27.09.2019 07:10


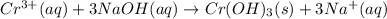
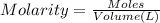
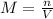
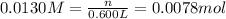
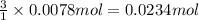 of NaOH
of NaOH