
Chemistry, 05.11.2019 04:31, angelapegues20097
A22.4 l high pressure reaction vessel is charged with 0.5020 mol of iron powder and 1.39 atm of oxygen gas at standard temperature. on heating, the iron and oxygen react according to the balanced reaction below. 4fe(s) + 3o2(g) → 2fe2o3(s) after the reaction vessel is cooled, and assuming the reaction goes to completion, what pressure of oxygen remains?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:10, hadellolo8839
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1

Do you know the correct answer?
A22.4 l high pressure reaction vessel is charged with 0.5020 mol of iron powder and 1.39 atm of oxyg...
Questions in other subjects:

Mathematics, 08.06.2021 19:00

Mathematics, 08.06.2021 19:00


English, 08.06.2021 19:00




Arts, 08.06.2021 19:00


Mathematics, 08.06.2021 19:10


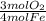 = 0.3765 mol O₂
= 0.3765 mol O₂



