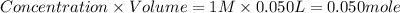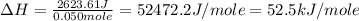Chemistry, 05.11.2019 02:31, briseno138
A50.0 ml sample of a 1.00 m solution of cuso4 is mixed with 50.0 ml of 2.00 m koh in a calorimeter. the temperature of both solutions was 20.2 degree celsious before mixing and 26.3 degree celsious after mixing . the heat capacity of the calorimeter is 12.1 j/k. from the data, calculate delta hfor the process
cuso4(1 m)+2koh(2 (oh)2(s)+k2so4(0.5 m)
assume that the specific heat and density of the solution after mixing are the same as those of pure water and that the volumes are additive.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, penny3109
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 00:20, paulawells11
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 07:00, misspicafunpoke
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2
Do you know the correct answer?
A50.0 ml sample of a 1.00 m solution of cuso4 is mixed with 50.0 ml of 2.00 m koh in a calorimeter....
Questions in other subjects:



Social Studies, 02.10.2019 17:40


Mathematics, 02.10.2019 17:40



Mathematics, 02.10.2019 17:40

History, 02.10.2019 17:40


![q=[q_1+q_2]](/tpl/images/0359/7670/341bc.png)
![q=[c_1\times \Delta T+m_2\times c_2\times \Delta T]](/tpl/images/0359/7670/1d50b.png)
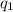 = heat absorbed by the calorimeter
= heat absorbed by the calorimeter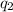 = heat absorbed by the solution
= heat absorbed by the solution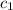 = specific heat of calorimeter =
= specific heat of calorimeter = 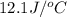
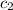 = specific heat of water =
= specific heat of water = 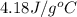
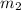 = mass of water or solution =
= mass of water or solution = 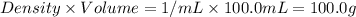
 = change in temperature =
= change in temperature = 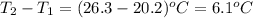
![q=[(12.1J/^oC\times 6.1^oC)+(100.0g\times 4.18J/g^oC\times 6.1^oC)]](/tpl/images/0359/7670/91dfe.png)
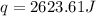
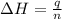
 = enthalpy change = ?
= enthalpy change = ?