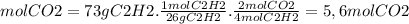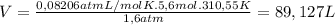Chemistry, 04.11.2019 22:31, kenleighbrooke67
Ethyne gas combusts with oxygen gas according to the following reaction: calculate the volume, in ml of co2 produced when 73 g of c2h2 react at 37.4 °c and 1.6 atm. (r = 0.08206 l atm/mol k) latex: 2\: c_2h_2\left(g\right)\: +\: 5o_2\left(g\right)\: \longrightarrow\: 4\: co_2\left(g\right)\: +2\: h_2o\left(l\right)\:

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:10, lilyjordan5972
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1

Chemistry, 22.06.2019 10:30, Wookas8355
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 11:30, claudr03
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3
Do you know the correct answer?
Ethyne gas combusts with oxygen gas according to the following reaction: calculate the volume, in m...
Questions in other subjects:







Biology, 30.10.2020 17:00


History, 30.10.2020 17:00