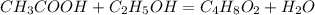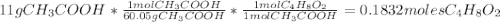Determine the theoretical maximum moles of ethyl acetate, , that could be produced in this experiment. the reactant, acetic acid, is the limiting reagent. (to avoid introducing rounding errors on intermediate calculations, enter your answer to four significant figures.) theoretical maximum moles of ethyl acetate = mol reactant mass 11.0 g product mass 10.8 g reactant moles 0.1833 mol

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, milesjreece3939
Select each correct answer. more than one answer may be correct. which of the following is a characteristic of unicellular organisms? they can possess tissues and organs. all of their functions are performed by a single cell. they are usually microscopic. each of their cells is specialized to perform a specific function.
Answers: 1

Chemistry, 22.06.2019 10:00, zionlopez543
Americium-241 undergoes fission to produce three neutrons per fission event. if a neutron-absorbing material is mixed in with this sample so that the rate of neutron production drops down to 1.8 neutrons per fission event, which will be effective at achieving a critical mass? check all that apply. remove a deflective shield surrounding the sample. remove absorbent material mixed in with the sample. compress the sample of americium-241.
Answers: 1

Chemistry, 22.06.2019 11:20, Jessicadiaz8602
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1
Do you know the correct answer?
Determine the theoretical maximum moles of ethyl acetate, , that could be produced in this experimen...
Questions in other subjects:


History, 21.01.2020 07:31



Mathematics, 21.01.2020 07:31


Health, 21.01.2020 07:31


Mathematics, 21.01.2020 07:31

Physics, 21.01.2020 07:31


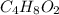 )
)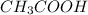 reacts with ethanol to produce ethyl acetate
reacts with ethanol to produce ethyl acetate