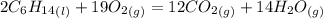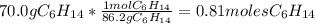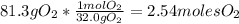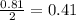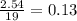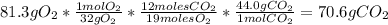Chemistry, 01.11.2019 02:31, twistedgamerhd12
Problem page liquid hexane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water . suppose 70. g of hexane is mixed with 81.3 g of oxygen. calculate the maximum mass of carbon dioxide that could be produced by the chemical reaction. be sure your answer has the correct number of significant digits.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 23:00, maddyleighanne
Arectangle has a diagonal 20 inches long that forms angles of 60 and 30 with the sides. find the perimeter of the rectangle. for geometry
Answers: 3

Chemistry, 23.06.2019 10:00, anonymous176
An uncovered pot of water lies out in the sun. which statements correctly describe what happens at the surface of the liquid water? 1. the vapor pressure remains constant regardless of the water temperature. 2. the vapor pressure is produced by water molecules that have evaporated. 3. the vapor pressure increases as the sun heats the water in the pot. 4. evaporation stops once the vapor pressure reaches a certain point. 5. evaporation and condensation both occur on the liquid’s surface.
Answers: 3
Do you know the correct answer?
Problem page liquid hexane will react with gaseous oxygen to produce gaseous carbon dioxide and gase...
Questions in other subjects:







Mathematics, 03.08.2019 11:30


Social Studies, 03.08.2019 11:30

Geography, 03.08.2019 11:30