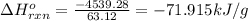Pentaborane−9 (b5h9) is a colorless, highly reactive liquid that will burst into flames when exposed to oxygen. the reaction is 2b5h9(l) + 12o2(g) → 5b2o3(s) + 9h2o(l) calculate the kilojoules of heat released per gram of the compound reacted with oxygen. the standard enthalpy of formations of b5h9(l), b2o3(s), and h2o(l) are 73.2, −1271.94, and −285.83 kj/mol, respectively.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, sethjohnson386pbnm3x
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 13:00, carlinryan
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 14:50, chem1014
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2
Do you know the correct answer?
Pentaborane−9 (b5h9) is a colorless, highly reactive liquid that will burst into flames when exposed...
Questions in other subjects:

Mathematics, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

English, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

Spanish, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

History, 16.09.2020 02:01


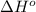
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_f(product)]-\sum [n\times \Delta H^o_f(reactant)]](/tpl/images/0352/3055/45485.png)
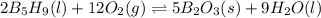
![\Delta H^o_{rxn}=[(n_{(B_2O_3)}\times \Delta H^o_f_{(B_2O_3)})+(n_{(H_2O)}\times \Delta H^o_f_{(H_2O)})]-[(n_{(B_5H_9)}\times \Delta H^o_f_{(B_5H_9)})+(n_{(O_2)}\times \Delta H^o_f_{(O_2)})]](/tpl/images/0352/3055/0889c.png)
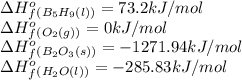
![\Delta H^o_{rxn}=[(5\times -1271.94)+(9\times -285.83)]-[(2\times 73.2)+(12\times 0)]=-9078.57kJ/mol](/tpl/images/0352/3055/ac167.png)
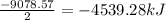
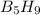 = 63.12 g/mole
= 63.12 g/mole