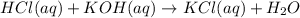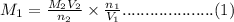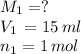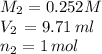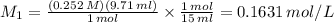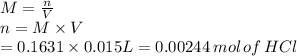1) if 9.71 ml of 0.252 m koh is used to completely titrate 1
or 0.252 m koh is used to complet...

Chemistry, 29.10.2019 23:31, cascyrio2002
1) if 9.71 ml of 0.252 m koh is used to completely titrate 1
or 0.252 m koh is used to completely titrate 15.0 ml of hci, how many moles of hcl are in
the solution? hint: start by drawing the balanced chemical equation.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, maddynichole2017
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1


Chemistry, 22.06.2019 16:50, TheOriginal2x
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 22.06.2019 21:30, sullivanjakob
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Social Studies, 14.04.2020 23:50


Engineering, 14.04.2020 23:50

Mathematics, 14.04.2020 23:50

Biology, 14.04.2020 23:50