
Chemistry, 25.10.2019 17:43, SuperWoman9172
Asample of gas contains 0.1900 mol of co(g) and 0.1900 mol of no(g) and occupies a volume of 22.0 l. the following reaction takes place: 2co(g) + 2no(g)2co2(g) + n2(g) calculate the volume of the sample after the reaction takes place, assuming that the temperature and the pressure remain constant.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:30, noathequeen
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 18:40, johnnysteeler9934
What is one real world example of a colligative property?
Answers: 2
Do you know the correct answer?
Asample of gas contains 0.1900 mol of co(g) and 0.1900 mol of no(g) and occupies a volume of 22.0 l....
Questions in other subjects:


Biology, 03.04.2020 15:31





Mathematics, 03.04.2020 15:32




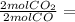 0.1900 mol CO₂
0.1900 mol CO₂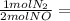 0.095 mol N₂
0.095 mol N₂



