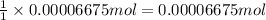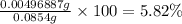Chemistry, 18.10.2019 22:00, hei40563273
In part 2 of the experiment, you will be analyzing a sample of household bleach. a 0.0854 g sample of household bleach is completely reacted with ki(s). the resulting solution is then titrated with 0.150 m nas2o3 solution. 0.890 ml of the solution is required to reach the colorless endpoint. what is the mass percent of naocl (mm = 74.44 g/mole) in the bleach? a. 30.1% b. 96.5% c. 2.23% d. 5.82%

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:00, daniel1480
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced. be sure your answer has the correct number of significant digits.
Answers: 2

Chemistry, 22.06.2019 18:00, brisacruz013
Which statement best describes the he properties of iconic compounds ?
Answers: 1

Chemistry, 22.06.2019 21:00, melissalopez12
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1
Do you know the correct answer?
In part 2 of the experiment, you will be analyzing a sample of household bleach. a 0.0854 g sample o...
Questions in other subjects:





English, 03.08.2019 15:30


Biology, 03.08.2019 15:30


Chemistry, 03.08.2019 15:30


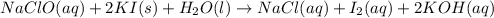 ..[1]
..[1]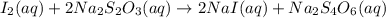 ..[2]
..[2]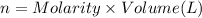
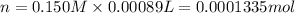
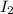 .
.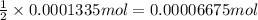 of
of