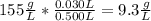Chemistry, 18.10.2019 21:00, pgjohnston001
Astudent placed 15.5 g of glucose (c6h12o6) in a volumetric flask, added enough water to dissolve the glucose by swirling, then carefully added additional water until the 100. ml mark on the neck of the flask was reached. the flask was then shaken until the solution was uniform. a 30.0 ml sample of this glucose solution was diluted to 0.500 l. how many grams of glucose are in 100. ml of the final solution?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, asims13
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 22.06.2019 22:30, angelagonzalesownus1
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1

Do you know the correct answer?
Astudent placed 15.5 g of glucose (c6h12o6) in a volumetric flask, added enough water to dissolve th...
Questions in other subjects:


English, 17.12.2021 14:00

Chemistry, 17.12.2021 14:00

Law, 17.12.2021 14:00

Chemistry, 17.12.2021 14:00


Biology, 17.12.2021 14:00

English, 17.12.2021 14:00


History, 17.12.2021 14:00