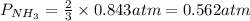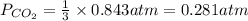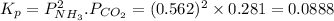Chemistry, 18.10.2019 19:10, daverius3153
7. nh2co2nh4(s) when heated to 450 k undergoes the following reaction to produce a system which reaches equilibrium: nh2co2nh4(s) ⇀↽ 2 nh3(g) + co2(g) the total pressure in the closed container under these condition is found to be 0.843 atm. calculate a value for the equilibrium constant, kp. a) 0.00701 b) 0.0888 c) 0.222 d) 0.599

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:10, aleilyg2005
Select the correct answer from each drop-down menu. describe what happens to a carbon-11 atom when it undergoes positron emission. the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 12:20, sindy35111
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l. s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3
Do you know the correct answer?
7. nh2co2nh4(s) when heated to 450 k undergoes the following reaction to produce a system which reac...
Questions in other subjects:










History, 15.11.2019 21:31


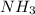 and
and 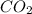 are gaseous. Hence equilibrium constant depends upon partial pressures of
are gaseous. Hence equilibrium constant depends upon partial pressures of 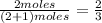
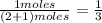
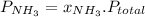 and P_{CO_{2}}= x_{CO_{2}}.P_{total}
and P_{CO_{2}}= x_{CO_{2}}.P_{total}