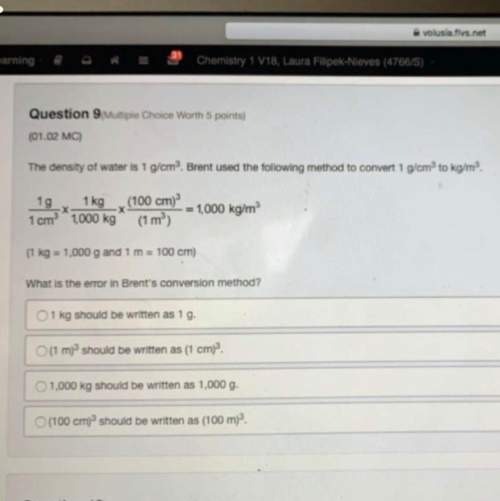
Which statement is true about the ionic size of the elements in a group as one moves from top to bottom in that group?
ionic size does not vary in any predictable way within the group.
ionic size increases from top to bottom within the group.
ionic size decreases from top to bottom within the group.
ionic size stays the same within the group.

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 08:30, myamiller558
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 10:30, jahmira96
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2
Do you know the correct answer?
Which statement is true about the ionic size of the elements in a group as one moves from top to bot...
Questions in other subjects:

History, 01.06.2020 20:59


Mathematics, 01.06.2020 20:59


Mathematics, 01.06.2020 20:59

History, 01.06.2020 20:59

Mathematics, 01.06.2020 20:59