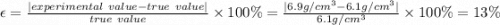Chemistry, 14.10.2019 22:00, yurlgurllmay
in a laboratory activity, the density of a sample of vanadium is determined to be 6.9 g/cm3 at room temperature. what is the percent error for the determined value?
a)
0.15%
b)
0.87%
c)
13%
d)
15%

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:20, pilarmonsivais
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3


Chemistry, 22.06.2019 15:30, lizzyhearts
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
Do you know the correct answer?
in a laboratory activity, the density of a sample of vanadium is determined to be 6.9 g/cm3 at room...
Questions in other subjects:

Physics, 16.09.2019 08:10

Mathematics, 16.09.2019 08:10

Mathematics, 16.09.2019 08:10

Mathematics, 16.09.2019 08:10


History, 16.09.2019 08:10

Biology, 16.09.2019 08:10

English, 16.09.2019 08:10

Computers and Technology, 16.09.2019 08:10

Social Studies, 16.09.2019 08:10