
Chemistry, 10.10.2019 02:00, thetudent41
Calculate the number of moles of al2o3 that are produced when 0.60 mol of fe is produced in the following reaction: 2al(s)+3feo(s) = 3fe(s)+al2o3(s)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, queenkimm26
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3


Chemistry, 22.06.2019 14:30, darkghostmist
What type of reaction fuels the processes seen here?
Answers: 2

Chemistry, 22.06.2019 21:00, andrethisman88
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1
Do you know the correct answer?
Calculate the number of moles of al2o3 that are produced when 0.60 mol of fe is produced in the foll...
Questions in other subjects:

English, 02.03.2021 20:10

Biology, 02.03.2021 20:10





Mathematics, 02.03.2021 20:10



English, 02.03.2021 20:10


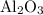 produced is
produced is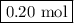 .
.
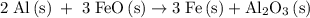
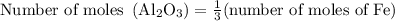 …… (1)
…… (1)
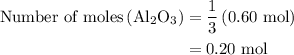
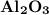 is produced when 0.60 mol of Fe is produced in the reaction.
is produced when 0.60 mol of Fe is produced in the reaction.



