
Chemistry, 09.10.2019 23:00, jakester00792
Which of the following reactions would be the most spontaneous at 298 k? which of the following reactions would be the most spontaneous at 298 k? a + b → c; e°cell = +1.22 v a + 2 b → c; e°cell = +0.98 v a + b → 3 c; e°cell = +0.15 v a + b → 2 c; e°cell = -0.030 v more information is needed to determine.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:50, Catracho3619
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1


Chemistry, 22.06.2019 09:10, aleilyg2005
Select the correct answer from each drop-down menu. describe what happens to a carbon-11 atom when it undergoes positron emission. the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 15:20, munziruddin204
Which description best characterizes the motion of particles in a solid?
Answers: 2
Do you know the correct answer?
Which of the following reactions would be the most spontaneous at 298 k? which of the following rea...
Questions in other subjects:

Mathematics, 09.05.2020 04:57




Computers and Technology, 09.05.2020 04:57

Mathematics, 09.05.2020 04:57

Social Studies, 09.05.2020 04:57



Engineering, 09.05.2020 04:57


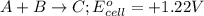
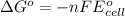
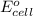 will be the most spontaneous.
will be the most spontaneous.



