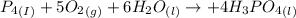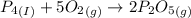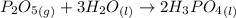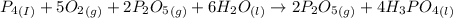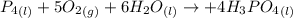Phosphoric acid, which is commonly used as rust inhibitor, food additive and etching agent for dental and orthopedic use, can be synthesized using a two-step thermal process. in the first step, phosphorus and oxygen react to form diphosphorus pentoxide: (l)(g)(g) in the second step, diphosphorus pentoxide and water react to form phosphoric acid: (g)(l)(l) write the net chemical equation for the production of phosphoric acid from phosphorus, oxygen and water. be sure your equation is balanced.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, EMQPWE
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1



Chemistry, 22.06.2019 12:00, winterblanco
What is the lowest number energy level where a d sublevel is found
Answers: 1
Do you know the correct answer?
Phosphoric acid, which is commonly used as rust inhibitor, food additive and etching agent for denta...
Questions in other subjects:



Mathematics, 25.06.2019 11:30

History, 25.06.2019 11:30

Physics, 25.06.2019 11:30

Mathematics, 25.06.2019 11:30


Mathematics, 25.06.2019 11:30

Mathematics, 25.06.2019 11:30

Health, 25.06.2019 11:30