Chemistry, 08.10.2019 22:20, murillokiara0kiki
Experiments with rats show that if rats are dosed with 3.0 mg/kg of cocaine (that is, 3.0 mg cocaine per kg of animal mass), the concentration of dopamine in their brains increases by 0.75 μm after 60 seconds. calculate how many molecules of dopamine would be produced in a rat (average brain volume 5.00 mm3) after 60 seconds of a 3.0 mg/kg dose of cocaine.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, mommatann
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 21.06.2019 23:30, lylessd423
Two atoms interact with each other as shown by the equation. complete the equation by filling in the missing parts. 1 2 3 4 5 h he li
Answers: 2

Chemistry, 22.06.2019 18:00, meowmeowcow
Find the mass, in grams, of 5.00*10^23 molecules of f2
Answers: 3
Do you know the correct answer?
Experiments with rats show that if rats are dosed with 3.0 mg/kg of cocaine (that is, 3.0 mg cocaine...
Questions in other subjects:


Mathematics, 19.09.2019 22:00

Chemistry, 19.09.2019 22:00


English, 19.09.2019 22:00




Social Studies, 19.09.2019 22:00

Mathematics, 19.09.2019 22:00


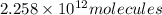 .
.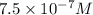
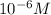
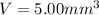
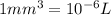
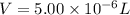
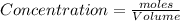
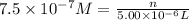
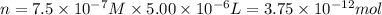
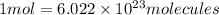
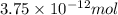 of dopamine :
of dopamine :