
Hydrogen sulfide (h2s) is a common and troublesome pollutant in industrial wastewaters. one way to remove h2s is to treat the water with chlorine, in which case the following reaction occurs: h2s(aq)+cl2(aq)→s(s)+2h+(aq)+2cl−(a q) the rate of this reaction is first order in each reactant. the rate constant for the disappearance of h2s at 28 ∘c is 3.5×10−2 m−1s−1.if at a given time the concentration of h2s is 2.0×10-4 m and that of cl2 is 2.8×10-2 m , what is the rate of formation of cl?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, tae8002001
How much energy moves onto the next level, in an energy pyramid
Answers: 1

Chemistry, 22.06.2019 12:30, poopybutt541
Avariable that is not being directly tested during an experiment should be
Answers: 1


Chemistry, 22.06.2019 18:00, kyllow5644
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3
Do you know the correct answer?
Hydrogen sulfide (h2s) is a common and troublesome pollutant in industrial wastewaters. one way to r...
Questions in other subjects:

Mathematics, 22.09.2020 08:01

World Languages, 22.09.2020 08:01


Mathematics, 22.09.2020 08:01

Biology, 22.09.2020 08:01


Mathematics, 22.09.2020 08:01


Mathematics, 22.09.2020 08:01

Mathematics, 22.09.2020 08:01



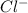 will be twice the rate of disappearance of
will be twice the rate of disappearance of  .
.![k[H_{2}S][Cl_{2}]](/tpl/images/0299/3822/2cbd6.png)
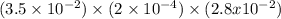
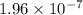 M/s
M/s

 M/s
M/s



